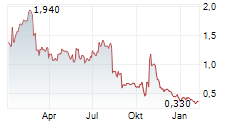
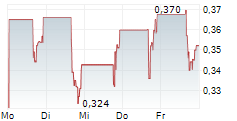
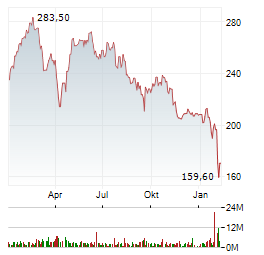
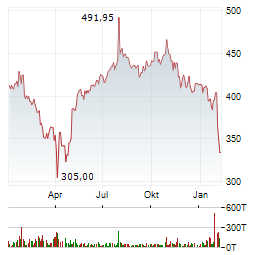
Chart-Typ:
Zeitraum:
| SAP Aktie: Kurssprung über den GD200 | Nach der angekündigten Zollpause ist die SAP-Aktie gestern mit einem Plus von 7,3% zurück über die 200-Tage-Linie gesprungen. Rückblick: Die SAP-Aktie war - ausgehend vom bisherigen Allzeithoch bei... ► Artikel lesen |
| DAX trotzt schwachen US-Vorgaben - Gold, SAP, Adidas, Apple, American Express, Tesla, Microsoft | Die Aktienmärkte weltweit pendeln derzeit zwischen den Extremen. Der DAX legte gestern über 4,5 Prozent zu. Die Vorgaben aus Übersee sind diesmal überwiegend schwach. Trotzdem deutet sich für den DAX... ► Artikel lesen |
| DAX höher, Gold mit Rekord: US-Banken, SAP, Deutsche Bank, Infineon, Rheinmetall, Telekom ... | Der DAX hat am Donnerstag zwischenzeitlich Kursgewinne von mehr als acht Prozent verzeichnen können. Dann ging ihm jedoch etwas die Luft aus. Am Ende schloss er 4,5 Prozent im Plus bei 20.562,73 Zählern.... ► Artikel lesen |
| Windows 11: Kontroverses Feature Recall soll endlich starten | Die Einführung von Microsofts Recall steht offenbar endgültig bevor. Gegen die KI-Funktion, die Screenshots von Aktivitäten auf Copilot Plus-PCs anfertigt, hatte es erhebliche Sicherheitsbedenken gegeben.... ► Artikel lesen |
| Aktie von Microsoft läuft heute schlechter (334,0562 €) | Am US-amerikanischen Aktienmarkt notiert das Wertpapier von Microsoft derzeit leichter. Der jüngste Kurs betrug 374,35 US-Dollar. Ein Kursverlust in Höhe von 15,58 US-Dollar müssen derzeit die Aktionäre... ► Artikel lesen |
| ROUNDUP/Aktien New York Schluss: Anleger treten wegen Zöllen auf die Bremse | NEW YORK (dpa-AFX) - Nach dem fulminanten Aufschwung vom Vortag haben die US-Aktienmärkte am Donnerstag wieder den Rückwärtsgang eingelegt. Der Dow Jones Industrial schloss 2,50 Prozent tiefer bei... ► Artikel lesen |
| CoreWeave: Insider casht aus - steigt Google ein? | Der Börsenneuling CoreWeave sorgt weiter für Schlagzeilen: Laut Medienberichten ist die Alphabet-Tochter Google an einem Leasing von Nvidia-Chips interessiert, die der KI-Hyper-Scaler im Bestand hat.... ► Artikel lesen |
| Nach Coreweave - nächstes Hammer-IPO? | Der USDC-Stablecoin-Emittent Circle Internet Financial hat ambitionierte Pläne: Am Dienstag reichte das Unternehmen ein Prospekt für einen Börsengang bei der US-Börsenaufsicht SEC ein. Damit positioniert... ► Artikel lesen |
| KI-Aktie explodiert: CoreWeave schießt 42 Prozent nach oben - Rallye oder Strohfeuer? | © Foto: Jakub Porzycki - NurPhotoDie Aktie von CoreWeave fährt Achterbahn. Kurz nach dem Börsengang rutschte sie unter den Ausgabepreis. Gestern schoss sie dann um 42 Prozent nach oben. Im vorbörslichen... ► Artikel lesen |
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und .
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu VSEE HEALTH INC finden Sie auf Wallstreet Online.