- Oral difelikefalin did not demonstrate meaningful clinical benefit compared to placebo -
- Company will discontinue clinical program in NP and explore strategic alternatives -
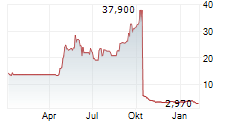
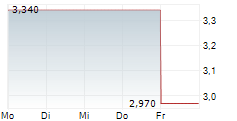
STAMFORD, Conn., June 12, 2024 (GLOBE NEWSWIRE) -- Cara Therapeutics, Inc. (Nasdaq: CARA), a development-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus, today announced the outcome from the dose-finding Part A of the KOURAGE-1 study evaluating the efficacy and safety of oral difelikefalin for moderate-to-severe pruritus in adult patients with notalgia paresthetica (NP). Oral difelikefalin did not demonstrate a meaningful clinical benefit at any dose compared to placebo, resulting in the Company's decision to discontinue the clinical program in NP.
"Given our strong proof-of-concept results in NP and the significant unmet need in this sensory neuropathy, we are disappointed that oral difelikefalin did not demonstrate a meaningful improvement in pruritus compared to placebo in the KOURAGE-1 Part A study," said Christopher Posner, President and Chief Executive Officer of Cara Therapeutics. "We are grateful for the patients and investigators who participated in this study. We will be winding down the Phase 2/3 clinical program in NP and exploring strategic alternatives focused on maximizing shareholder value."
KOURAGE-1 Part A was a multicenter, randomized, double-blind, placebo-controlled study designed to inform the dose and sample size for the pivotal portions of the Phase 2/3 clinical program. In Part A, 214 patients were randomized to one of four arms: oral difelikefalin 2 mg twice a day (BID), 1 mg BID, 0.25 mg BID or placebo BID for 8 weeks. The primary endpoint was the proportion of patients achieving a =4-point improvement from baseline in the weekly mean of the daily 24-hour Itch-Numeric Rating Scale (I-NRS) score at Week 8.
Oral difelikefalin did not demonstrate a meaningful clinical benefit at any dose compared to placebo. The drug was generally well tolerated with a safety profile similar to prior trials.
As of March 31, 2024, the Company had approximately $70 million in cash, cash equivalents, and marketable securities.
About Cara Therapeutics
Cara Therapeutics is a development-stage biopharmaceutical company leading a new treatment paradigm to improve the lives of patients suffering from pruritus. The Company developed an IV formulation of difelikefalin, which is approved in the United States, EU, and multiple other countries for the treatment of moderate-to-severe pruritus associated with advanced chronic kidney disease in adults undergoing hemodialysis. The IV formulation is out-licensed worldwide. For more information, visit www.CaraTherapeutics.com and follow the company on X (Twitter), LinkedIn and Instagram.
Forward-looking Statements
Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Examples of these forward-looking statements include statements concerning the winding down of the Company's Phase 2/3 clinical program in NP, the exploration of strategic alternatives and other statements that are not historical facts. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. These risks are described more fully in Cara Therapeutics' filings with the Securities and Exchange Commission, including the "Risk Factors" section of the Company's Annual Report on Form 10-K for the year ending December 31, 2023 and its other documents subsequently filed with or furnished to the Securities and Exchange Commission, including its Form 10-Q for the quarter ended March 31, 2024. All forward-looking statements contained in this press release speak only as of the date on which they were made. Cara Therapeutics undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
MEDIA CONTACT:
Annie Spinetta
6 Degrees
973-768-2170
aspinetta@6degreespr.com
INVESTOR CONTACT:
Iris Francesconi, Ph.D.
Cara Therapeutics
203-406-3700
investor@caratherapeutics.com