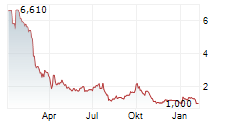
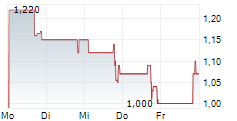
TEL AVIV, Israel and RALEIGH, N.C., Aug. 16, 2024 /PRNewswire/ -- RedHill Biopharma Ltd. (Nasdaq: RDHL) ("RedHill" or the "Company"), a specialty biopharmaceutical company, today announced that it plans to implement a ratio change of the Company's American Depositary Shares (ADSs) to its non-traded ordinary shares from the current ratio of one (1) ADS representing four hundred (400) ordinary shares to a new ratio of one (1) ADS representing ten thousand (10,000) ordinary shares. The anticipated first date of the ratio change is on or about August 20, 2024, and the Company's ADSs will continue to be traded on the Nasdaq Capital Market ("Nasdaq") under the symbol "RDHL" with a new CUSIP Number 757468301.
For ADS holders, the ratio change will have the same effect as a one-for-25 reverse ADS split. No new ADSs will be issued in connection with the change in the ADS ratio. ADS holders will not be required to take any action and, on the effective date, the Bank of New York Mellon, the depositary bank for RedHill's ADS program, will arrange for the exchange of every twenty-five (25) existing ADSs held for one (1) new ADS.
No fractional new ADSs will be issued in connection with the change in the ADS ratio. Instead, fractional entitlements to new ADSs will be aggregated, and the depositary bank will attempt to sell them with the net cash proceeds from the sale of the fractional ADS entitlements to be distributed to the applicable ADS holders by the depositary bank.
On March 14, 2024, the Company reported that it had received notification from the Nasdaq Capital Market, LLC that it did not comply with the minimum bid price requirements set forth in Nasdaq Listing Rule 5450(a)(2) for continued listing on Nasdaq. The Company anticipates that after the ratio change, the price of its ADSs will increase proportionally and meet the Nasdaq minimum bid price requirement, however there can be no assurance that such an increase will occur.
About RedHill Biopharma
RedHill Biopharma Ltd. (Nasdaq: RDHL) is a specialty biopharmaceutical company primarily focused on gastrointestinal and infectious diseases. RedHill promotes the gastrointestinal drugs Talicia®, for the treatment of Helicobacter pylori (H. pylori) infection in adults[i], and Aemcolo®, for the treatment of travelers' diarrhea in adults[ii]. RedHill's key clinical late-stage development programs include: (i) opaganib (ABC294640), a first-in-class oral broad-acting, host-directed SPHK2 selective inhibitor with potential for pandemic preparedness, targeting multiple indications with a U.S. government collaboration for development for Acute Radiation Syndrome (ARS), a Phase 2/3 program for hospitalized COVID-19, and a Phase 2 program in oncology; (ii) RHB-107 (upamostat), an oral broad-acting, host-directed, serine protease inhibitor with potential for pandemic preparedness is in late-stage development as a treatment for non-hospitalized symptomatic COVID-19, with non-dilutive external funding covering the entirety of the RHB-107 arm of the 300-patient Phase 2 adaptive platform trial, and is also targeting multiple other cancer and inflammatory gastrointestinal diseases; (iii) RHB-102, with potential UK submission for chemotherapy and radiotherapy induced nausea and vomiting, positive results from a Phase 3 study for acute gastroenteritis and gastritis and positive results from a Phase 2 study for IBS-D; (iv) RHB-104, with positive results from a first Phase 3 study for Crohn's disease; and (v) RHB-204, a Phase 3-stage program for pulmonary nontuberculous mycobacteria (NTM) disease.
More information about the Company is available at www.redhillbio.com / X.com/redhillbio.
Forward-Looking Statements
This press release contains "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995 and may discuss investment opportunities, stock analysis, financial performance, investor relations, and market trends. Such statements, including, but not limited to, statements regarding the potential effects from the ratio change, may be preceded by the words "intends," "may," "will," "plans," "expects," "anticipates," "projects," "predicts," "estimates," "aims," "believes," "hopes," "potential" or similar words and include statements regarding the risk that the Company will not comply with the listing requirements of Nasdaq to remain listed for trading on Nasdaq, timing of opaganib's development for Acute Radiation Syndrome, non-dilutive development funding from RHB-107 and its inclusion in a key platform study. Forward-looking statements are based on certain assumptions and are subject to various known and unknown risks and uncertainties, many of which are beyond the Company's control and cannot be predicted or quantified, and consequently, actual results may differ materially from those expressed or implied by such forward-looking statements. Such risks and uncertainties include, without limitation: market and other conditions; the timing, consummation and impact of the ratio change; the Company's ability to regain compliance with Nasdaq's minimum bid price requirements; the risk that the addition of new revenue generating products or out-licensing transactions will not occur; the risk that acceptance onto the RNCP Product Development Pipeline will not guarantee ongoing development or that any such development will not be completed or successful; the risk that the FDA does not agree with the Company's proposed development plans for opaganib for any indication, the risk that observations from preclinical studies are not indicative or predictive of results in clinical trials; the risk that the FDA pre-study requirements will not be met and/or that the Phase 3 study of RHB-107 in COVID-19 outpatients will not be approved to commence or if approved, will not be completed or, should that be the case, that we will not be successful in obtaining alternative non-dilutive development funding for RHB-107, the risk that HB-107's late-stage development for non-hospitalized COVID-19 will not benefit from the resources redirected from the terminated RHB-204 Phase 3 study, that the Phase 2/3 COVID-19 study for RHB-107 may not be successful and, even if successful, such studies and results may not be sufficient for regulatory applications, including emergency use or marketing applications, and that additional COVID-19 studies for opaganib and RHB-107 are likely to be required, as well as risks and uncertainties associated with the risk that the Company will not successfully commercialize its products; as well as risks and uncertainties associated with (i) the initiation, timing, progress and results of the Company's research, manufacturing, pre-clinical studies, clinical trials, and other therapeutic candidate development efforts, and the timing of the commercial launch of its commercial products and ones it may acquire or develop in the future; (ii) the Company's ability to advance its therapeutic candidates into clinical trials or to successfully complete its pre-clinical studies or clinical trials or the development of a commercial companion diagnostic for the detection of MAP; (iii) the extent and number and type of additional studies that the Company may be required to conduct and the Company's receipt of regulatory approvals for its therapeutic candidates, and the timing of other regulatory filings, approvals and feedback; (iv) the manufacturing, clinical development, commercialization, and market acceptance of the Company's therapeutic candidates and Talicia®; (v) the Company's ability to successfully commercialize and promote Talicia® and Aemcolo®; (vi) the Company's ability to establish and maintain corporate collaborations; (vii) the Company's ability to acquire products approved for marketing in the U.S. that achieve commercial success and build its own marketing and commercialization capabilities; (viii) the interpretation of the properties and characteristics of the Company's therapeutic candidates and the results obtained with its therapeutic candidates in research, pre-clinical studies or clinical trials; (ix) the implementation of the Company's business model, strategic plans for its business and therapeutic candidates; (x) the scope of protection the Company is able to establish and maintain for intellectual property rights covering its therapeutic candidates and its ability to operate its business without infringing the intellectual property rights of others; (xi) parties from whom the Company licenses its intellectual property defaulting in their obligations to the Company; (xii) estimates of the Company's expenses, future revenues, capital requirements and needs for additional financing; (xiii) the effect of patients suffering adverse experiences using investigative drugs under the Company's Expanded Access Program; (xiv) competition from other companies and technologies within the Company's industry; and (xv) the hiring and employment commencement date of executive managers. More detailed information about the Company and the risk factors that may affect the realization of forward-looking statements is set forth in the Company's filings with the Securities and Exchange Commission (SEC), including the Company's Annual Report on Form 20-F filed with the SEC on April 8, 2024. All forward-looking statements included in this press release are made only as of the date of this press release. The Company assumes no obligation to update any written or oral forward-looking statement, whether as a result of new information, future events or otherwise unless required by law.
Category: Financials
i. Talicia® (omeprazole magnesium, amoxicillin and rifabutin) is indicated for the treatment of H. pylori infection in adults. For full prescribing information see: www.Talicia.com.
ii. Aemcolo® (rifamycin) is indicated for the treatment of travelers' diarrhea caused by noninvasive strains of Escherichia coli in adults. For full prescribing information see: www.Aemcolo.com.
Logo: https://mma.prnewswire.com/media/1334141/RedHill_Biopharma_Logo.jpg
Company contact:
Adi Frish
Chief Corporate and Business Development Officer
RedHill Biopharma
+972-54-6543-112
[email protected]
SOURCE RedHill Biopharma Ltd.