NEW YORK CITY (dpa-AFX) - Bristol-Myers Squibb Company (BMY), Monday announced that the European Medicines Agency has validated its Type II variation application to expand the indication for Breyanzi for the treatment of adult patients with relapsed or refractory follicular lymphoma or FL who have received two or more prior lines of systemic therapy.
The CAR T Cell therapy Breyanzi is made from a patient's own T cells, which are collected and genetically reengineered to become CAR T cells that are then delivered via infusion as a one-time treatment, the company added.
The application is backed by data from the Phase 2 TRANSCEND FL study, where Breyanzi demonstrated a high overall response rate in adults with relapsed or refractory FL.
Additionally, Japan's Ministry of Health, Labour and Welfare has approved a supplemental New Drug Application for Breyanzi for the treatment of relapsed or refractory FL after one prior line of systemic therapy in patients with high-risk FL and after two or more lines of systemic therapy, making it the first CAR T approved in the second-line setting anywhere and the third approval for Breyanzi in Japan.
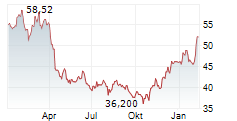
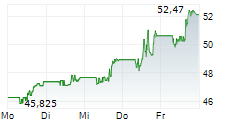
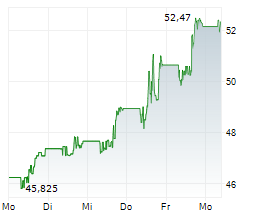
Currently, Bristol-Myers Squibb's stock is trading at $49.82, up 0.89 percent on the New York Stock Exchange.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News