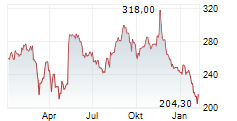
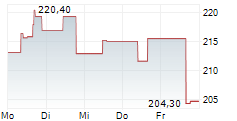
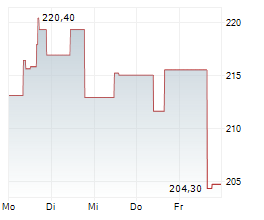
WASHINGTON (dpa-AFX) - Insulet Corp. (PODD) has just received the green light from the U.S. Food and Drug Administration for its groundbreaking SmartAdjust Omnipod 5 automated insulin delivery system, tailored for adults aged 18 and above with Type 2 diabetes.
This landmark approval represents the first-ever device specifically crafted for automated insulin dosing for individuals with Type 2 diabetes. The Omnipod is a cutting-edge, tubeless wearable device capable of providing continuous insulin delivery for up to 72 hours without the need for traditional needles.
What sets the Omnipod 5 apart is its integration of a continuous glucose monitor, empowering users to manage their blood sugar levels without the inconvenience of daily injections or finger pricks. Notably, it can be operated through a compatible smartphone or a separate controller within the U.S.
Jim Hollingshead, President and CEO of Insulet, expressed his excitement about the potential impact of the Omnipod 5, stating, 'Omnipod 5 is setting a new standard in diabetes management, and we are thrilled with the opportunity to make a lasting impact on the insulin-requiring type 2 diabetes community.'
Before granting approval, the FDA rigorously evaluated data from a comprehensive clinical study involving 289 adults with Type 2 diabetes, all of whom were already using insulin and had utilized the SmartAdjust technology for a duration of 13 weeks.
The study encompassed participants with varied experiences in managing insulin and diabetes, including those on prevalent diabetes medications like GLP1 agonists. Results indicated notably improved blood sugar control across all demographic groups. On average, participants experienced a reduction in HbA1c levels of 0.8 percentage points, while those with HbA1c levels of 9.0% or higher saw a significant decrease of 2.1 points. Throughout the study, no serious adverse events or complications were reported, with only mild to moderate side effects observed, including instances of low and high blood sugar, as well as skin irritation.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News