PARIS (dpa-AFX) - Regeneron Pharmaceuticals, Inc. (REGN) and Sanofi (SNY), Friday announced that the U.S. Food and Drug Administration has approved Dupixent as an add-on maintenance treatment of adults with inadequately controlled chronic obstructive pulmonary disease or COPD and an eosinophilic phenotype. Sanofi claimed that Dupixent will be nation's first biologic medicine approved for these conditions.
COPD is a respiratory disease that damages the lungs and causes progressive lung function decline. Its symptoms include persistent cough, excessive mucus production, and shortness of breath.
The approval was based on phase 3 studies that demonstrated reduction in exacerbations, as well as improvements in lung function and health-related quality of life in Dupixent patients compared to placebo.
In July, Dupixent received approval from the European Medicines Agency as an add-on maintenance treatment for adults with uncontrolled COPD characterized by raised blood eosinophils.
Dupixent is awaiting regulatory approvals in several nations across the world, including in Japan, Sanofi noted.
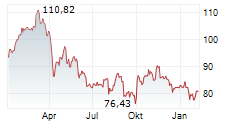
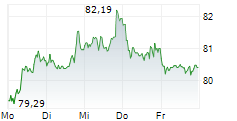
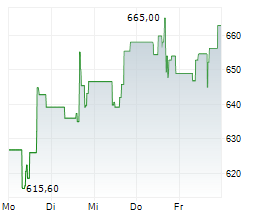
Currently, Regeneron's stock is trading at $1,063.05, up 2.22 percent, and Sanofi's stock is trading at $57.90, up 1.60 percent on the Nasdaq.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News