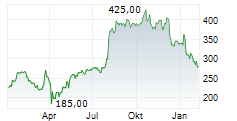
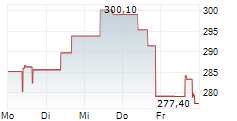
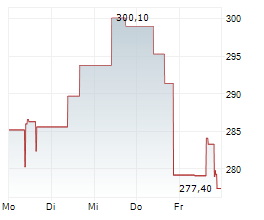
BEIJING (dpa-AFX) - Alnylam Pharmaceuticals Inc. (ALNY) said that new data from the HELIOS-B Phase 3 study of vutrisiran showed that the drug attenuated measures of disease progression across multiple domains of cardiac structure and function, NT-proBNP and troponin I, in a contemporary ATTR amyloidosis with cardiomyopathy patient population.
The data demonstrated that rapid knockdown of TTR leads to an early impact on cardiac biomarkers and echocardiographic parameters, indicative of a potential disease-modifying effect, and underscores the benefit of treating patients with an RNAi therapeutic earlier in the course of disease.
The company said it remains confident that, with approval, vutrisiran has the potential to become a first-line therapy for ATTR amyloidosis with cardiomyopathy and is on track to complete multiple global regulatory submissions before the end of the year.
vutrisiran is an investigational RNAi therapeutic in development for the treatment of ATTR amyloidosis with cardiomyopathy (ATTR-CM).
According to the company, the new echocardiographic data demonstrated that treatment with vutrisiran slowed disease progression in a contemporary population of patients with ATTR-CM across multiple domains of cardiac structure and diastolic and systolic function at Month 30 as compared to placebo. The magnitude of the treatment effects with vutrisiran compared to placebo were similar or greater in the monotherapy population. Significant improvements in both diastolic and systolic function were observed as early as 12 months and 18 months, respectively, in the overall population.
At Month 30, the relative reduction in the fold change in NT-proBNP in patients treated with vutrisiran compared to placebo was 32% in the overall population and 43% in the monotherapy population. At Month 30, relative reduction in the fold change of troponin I was 32% in the overall population and 45% in the monotherapy population.
In the subgroup of patients receiving tafamidis at baseline, a relative reduction in the fold change of 18% was observed in NT-proBNP and 10% in troponin I at Month 30. For both NTproBNP and troponin I, patients treated with vutrisiran demonstrated nominally significant reductions relative to placebo at 6 months.
For More Such Health News, visit rttnews.com.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News