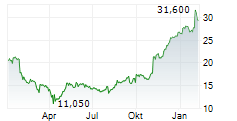
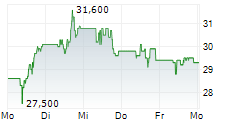
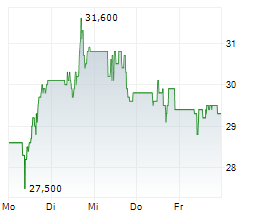
PETAH TIKVA (dpa-AFX) - Teva Pharmaceutical Industries (TEVA) announced the FDA has accepted, and the European Medicines Agency has validated, applications for TVB-009P, a biosimilar candidate to Prolia. The FDA's decision and EMA's expected opinion are anticipated in the second half of 2025.
Teva said, TVB-009P, proposed biosimilar to Prolia, is the first of Teva's internally developed biosimilars to be submitted to the U.S. FDA. Both applications include all indications approved for the reference product, Prolia, including conditions with a high risk for fracture, such as osteoporosis in postmenopausal women. TVB-009P is an investigational product and has not received regulatory approval in any country.
For More Such Health News, visit rttnews.com.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News