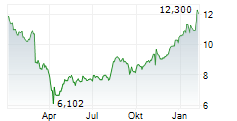
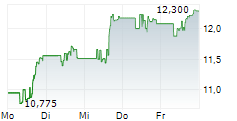
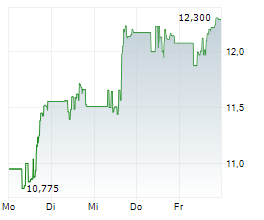
WASHINGTON (dpa-AFX) - Viatris (VTRS) reported positive top-line results of Phase 3 study in Japan evaluating the safety and efficacy of EFFEXOR in adults with generalized anxiety disorder. The study achieved its primary objective of superiority of anxiolytic effects of venlafaxine compared to placebo at 8 weeks. All seven secondary efficacy endpoints were met, which confirmed superiority compared to placebo. In the study, EFFEXOR was generally well tolerated. The company expects to present the full results from this Phase 3 study at a future medical congress.
EFFEXOR is currently approved in Japan for the indication of major depressive disorder in adults. EFFEXOR has also been approved for the indication of GAD in more than 80 countries outside of Japan.
For More Such Health News, visit rttnews.com.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News