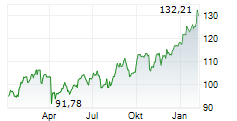
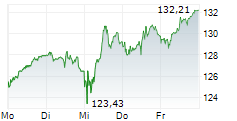
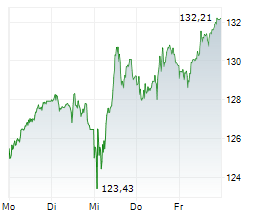
BASEL (dpa-AFX) - Novartis (NVS) presented 12-month data from the Phase III APPEAR-C3G study at the American Society of Nephrology or ASN Kidney Week 2024. The results showed that patients with C3 glomerulopathy (C3G) treated with oral Fabhalta (iptacopan) alongside supportive care experienced clinically meaningful and sustained results over one year.
The data confirmed that treatment with Fabhalta led to significant proteinuria reduction, observable as early as 14 days and maintained at 12 months. Additionally, during the open-label phase of the study, participants who switched to Fabhalta also showed a reduction in proteinuria. Improvement in the estimated glomerular filtration rate (eGFR) slope was noted upon initiation of Fabhalta, compared to patients' historic rapid decline, based on results from a prespecified exploratory analysis.
The company stated that Fabhalta exhibited a favorable safety profile, with no new safety signals identified.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News