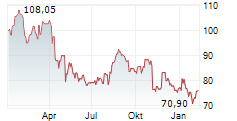
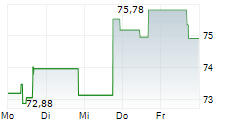
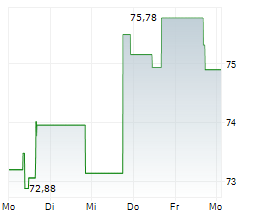
WASHINGTON (dpa-AFX) - Zimmer Biomet (ZBH) announced FDA 510(k) clearance of Persona SoluTion Porous Plasma Spray Femur, a total knee implant component offering an alternative for patients with sensitivities to bone cement and/or metal. Persona SoluTion PPS Femur will be commercially available in the U.S. in first quarter, 2025.
Joe Urban, President, Knees at Zimmer Biomet, said: 'Persona SoluTion PPS Femur combines our latest advances in cementless fixation with decades of proprietary clinical expertise in developing novel materials and surface hardening processes.'
The Persona SoluTion PPS Femur features a porous coating for cementless fixation and leverages a surface treatment designed to enhance wear performance.
For More Such Health News, visit rttnews.com.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News