WASHINGTON (dpa-AFX) - Neurocrine Biosciences, Inc. (NBIX), Friday announced the commercial launch of Crenessity in the U.S. for adult and pediatric patients with classic congenital adrenal hyperplasia or CAH, a rare genetic condition.
The announcement follows the U.S. Food and Drug Administration's approval of Crenessity as an adjunctive treatment to glucocorticoid replacement to control androgens in CAH patients.
The biopharmaceutical company added that the medication will be exclusively available through PANTHERx Rare, a specialty pharmacy.
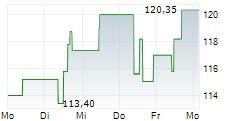
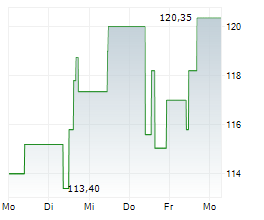
During the pre-market hours, Neurocrine's stock is trading at $134.01, down 0.70 percent on the Nasdaq.
Copyright(c) 2024 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2024 AFX News