WASHINGTON (dpa-AFX) - Teleflex (TFX) announced FDA 510(k) clearance of the AC3 Range Intra-Aortic Balloon Pump. The AC3 Range IABP is designed to provide ongoing IABP support across various patient transport modes, including ambulances and both fixed- and rotary-wing aircrafts. The AC3 Range IABP will enter full market release in the United States and will begin shipping to customers in the second quarter of 2025.
The company said its AC3 Range IABP combines the simple interface and algorithms of the AC3 Optimus IABP to deliver the same precisely timed support with features designed specifically for challenges unique to transport, including a full-size helium tank, dual power options, a metal-reinforced extendable handle, and four 360-degree swivel wheels for maneuverability.
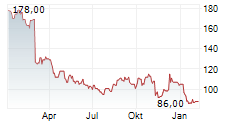
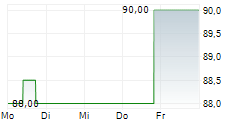
Shares of Teleflex are down 0.5% in pre-market trade on Tuesday.
For More Such Health News, visit rttnews.com.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News