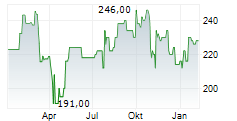
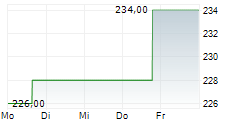
WASHINGTON (dpa-AFX) - Labcorp (LH), Tuesday announced it will offer nationwide a U.S. Food and Drug Administration (FDA) approved human papillomavirus (HPV) self-collection solution that can help women and their physicians asses the risk of developing cervical cancer.
Labcorp will also offer a vaginal swab self-collect option for sexually transmitted infection (STI) testing. Both solutions are available by physician order and can be completed privately in a healthcare setting or at any one of Labcorp's 2,200 Patient Service Centers (PSCs) nationwide.
'Many patients delay or avoid necessary health screenings due to discomfort, stigma or time challenges, which can lead to health complications and further spread infections,' said Dr. Brian Caveney, Labcorp's chief medical and scientific officer. 'Labcorp is now offering self-collection at our PSCs and healthcare settings such as physician offices to break down these barriers. With convenient access to essential tests and screenings, patients can take control of their health, on their own terms, when it's most convenient for them.'
Each year in the U.S. more than 13,000 women are diagnosed with cervical cancer and approximately 4,000 die from this preventable disease. HPV testing is a crucial tool for preventing cervical cancer through early intervention and treatment.
According to the U.S. Centers for Disease Control and Prevention (CDC), one in four women of screening age were not up to date with cervical cancer screening recommendations in 2021.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News