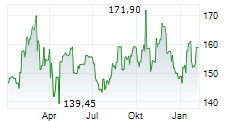
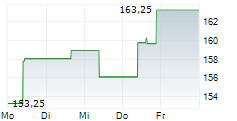
WASHINGTON (dpa-AFX) - Quest Diagnostics (DGX), a diagnostic services provider, announced Wednesday a new FDA-cleared human papillomavirus (HPV) self-collection solution aimed at broadening access to HPV screening to help identify women who are at risk of developing cervical cancer.
Physicians can now offer patients the option to collect their own specimen for HPV screening in a physician's office or other healthcare setting, with goal to reduce barriers to screening by enabling discreet self-collection.
The company also plans to introduce the self-collection option for patients, with a physician's order, at its 2,000 Quest patient service centers nationwide early next month.
The new option builds on Quest's experience in providing patients with discrete self-collection options.
The new offering utilizes the FDA-cleared HPV self-collection solution from Roche (RHHBY), approved for use with Roche's cobas HPV test in May 2024. Interested patients can opt for self-collection in clinical settings, such as a doctor's office, or other healthcare settings.
The solution is not FDA approved for at-home collection, and self-collection is not intended to replace a pelvic examination provided by a clinician.
The company had introduced a self-collection service option for vaginitis and other genital tract infections, such as chlamydia, gonorrhea, trichomoniasis and Mycoplasma genitalium, at its patient service centers in October 2024.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News