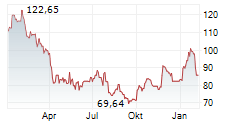
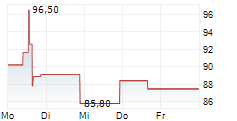
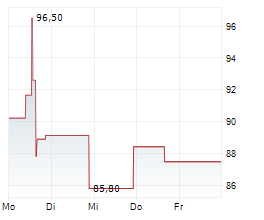
WASHINGTON (dpa-AFX) - Revvity, Inc. (RVTY), Wednesday said that the U.S. Food and Drug Administration (FDA) has approved the Auto-Pure 2400 liquid handling platform with the T-SPOT. tuberculosis (TB) test.
'The integration of the Auto-Pure 2400 platform with the T-SPOT.TB test gives laboratories the ability to process latent TB tests at higher volumes without compromising clinical accuracy,' said Yves Dubaquie, senior vice president, diagnostics at Revvity.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News