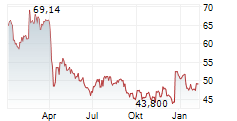
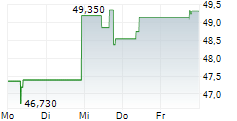
WASHINGTON (dpa-AFX) - BioMarin Pharmaceutical Inc. (BMRN) Wednesday said its Phase 3 PEGASUS study evaluating Palynziq in adolescents aged 12-17 with phenylketonuria (PKU) met its primary goal.
The study is designed to evaluate the safety and efficacy of Palynziq compared to diet alone in 55 adolescents aged 12-17 with phenylketonuria. The primary endpoints are change in blood Phe concentration and characterization of the safety profile in adolescents.
The drug is already approved to reduce blood Phe concentrations for adults in the U.S., for people 16 and older in the EU, Canada and Brazil, and for people 15 and older in Japan with PKU.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News