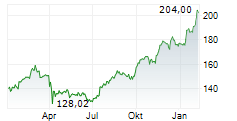
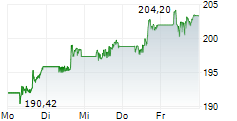
NEW BRUNSWICK (dpa-AFX) - Drug major Johnson & Johnson (JNJ) announced Friday positive results from the TREMFYA (guselkumab) Phase 3b APEX study in adults living with active psoriatic arthritis or PsA, compared to placebo.
The company noted that TREMFYA demonstrated clinically meaningful and statistically significant efficacy in patients with active PsA at risk for structural damage in Phase 3b study.
PsA is a chronic, immune-mediated, inflammatory disease characterized by peripheral joint inflammation, enthesitis, dactylitis, axial disease and the skin lesions associated with plaque psoriasis.
Johnson & Johnson's TREMFYA, a prescription medicine, is the first approved fully-human, dual-acting monoclonal antibody designed to neutralize inflammation at the cellular source.
APEX is a multicenter, randomized, double-blind, placebo-controlled study in patients with active PsA who are biologic naïve and have had an inadequate response to standard therapies.
Johnson & Johnson said the trial achieved both its primary endpoint (ACR20a) of reducing signs and symptoms and its major secondary endpoint of reducing progression of structural damage as measured by radiographic progression at 24 weeks in PsA compared to placebo.
The company noted that TREMFYA (guselkumab) is the first and only IL-23 inhibitor to significantly reduce both the signs and symptoms and the progression of structural damage in adults living with active psoriatic arthritis. Results from the APEX study are being prepared for presentation at upcoming medical congresses.
Terence Rooney, Vice President, Rheumatology Disease Area Leader, Johnson & Johnson Innovative Medicine, said, 'Psoriatic arthritis can be a progressive and debilitating disease, and without early identification and treatment, patients may experience irreversible joint damage that significantly impacts their daily activities. These new topline data highlight the importance of addressing both inflammation and structural damage at the source as early as possible. As the only IL-23 treatment to show significant inhibition of structural damage, TREMFYA equips healthcare providers with critical data so their patients do not have to compromise their future joint health.'
TREMFYA is approved in the U.S. to treat adults with moderate to severe plaque psoriasis, active psoriatic arthritis, moderately to severely active ulcerative colitis, and moderately to severely active Crohn's disease.
Europe, Canada, Japan, and a number of other countries have approved TREMFYA for the treatment of adults with moderate-to-severe plaque psoriasis and for the treatment of adults with active psoriatic arthritis.
For More Such Health News, visit rttnews.com
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News