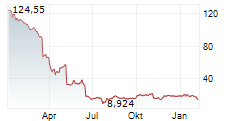
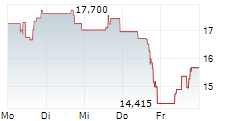
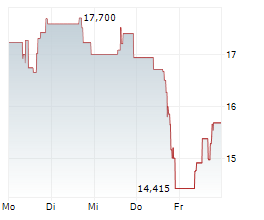
WASHINGTON (dpa-AFX) - Sarepta Therapeutics (SRPT) issued an update related to ELEVIDYS, the only approved gene therapy in patients with Duchenne muscular dystrophy. Following the safety update on acute liver failure that was issued on March 18, European Union reference member country authorities requested that the independent data monitoring committee meet to review the adverse event. The independent DMC met on April 3 and concurred that based on the totality of evidence, the overall benefit-risk profile remains favorable to continue dosing in the paused clinical trials without changes to the study protocols. Sarepta and Roche will submit this information in a response to the temporary halt within a week.
ELEVIDYS is indicated for the treatment of Duchenne muscular dystrophy in individuals at least 4 years of age.
Shares of Sarepta Therapeutics are down 1% in pre-market trade on Friday.
For More Such Health News, visit rttnews.com.
Copyright(c) 2025 RTTNews.com. All Rights Reserved
Copyright RTT News/dpa-AFX
© 2025 AFX News