- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 21.01. | Actuate Therapeutics plans phase 1/2 trial of oral cancer drug | 2 | Investing.com | ||
| 21.01. | Actuate Therapeutics Announces Plans to Expand Clinical Pipeline, Advancing Elraglusib Tablet into a Phase 1/2 Clinical Program in Refractory Cancers | 173 | GlobeNewswire (Europe) | -Initiation of phase 1 portion of the trial planned in 2H 2026 -Phase 2 portion of the trial will initiate development of the elraglusib tablet in specific indications, including refractory melanoma... ► Artikel lesen | |
| 15.01. | H.C. Wainwright reiterates Buy rating on Actuate Therapeutics stock at $20 | 2 | Investing.com | ||
| 12.01. | ACTUATE THERAPEUTICS, INC. - 8-K, Current Report | - | SEC Filings | ||
| 12.01. | Actuate Says Elraglusib Combo Boosts Survival In Metastatic Pancreatic Cancer | 2 | RTTNews | ||
| 12.01. | Actuate Therapeutics Reports Positive Follow-Up Data from its Randomized Controlled Phase 2 Trial Demonstrating Extended Long-Term Overall Survival Benefit with Elraglusib Plus Chemotherapy for Metastatic Pancreatic Cancer in Oral Presentation ... | 118 | GlobeNewswire (Europe) | Phase 2 study met its primary endpoint, demonstrating statistically significant improved overall survival with elraglusib plus gemcitabine/nab-paclitaxel versus chemotherapy aloneElraglusib combination... ► Artikel lesen | |
| ACTUATE THERAPEUTICS Aktie jetzt für 0€ handeln | |||||
| 06.01. | Actuate Therapeutics stock gets Buy rating after positive Phase 1 results | 4 | Investing.com | ||
| 06.01. | Lucid Capital Markets bestätigt Kaufempfehlung für Actuate Therapeutics nach Studienerfolg | 5 | Investing.com Deutsch | ||
| 06.01. | Actuate Therapeutics: Aktie steigt nach vielversprechenden Studiendaten zu Krebsmedikament für Kinder | 2 | Investing.com Deutsch | ||
| 06.01. | Actuate Therapeutics meldet positive Studienergebnisse bei Krebstherapie für Kinder | 2 | Investing.com Deutsch | ||
| 06.01. | Actuate reports positive results in pediatric cancer treatment trial | 1 | Investing.com | ||
| 06.01. | Actuate Therapeutics Announces Positive Patient Outcomes from Phase 1 Trial in Difficult-to-Treat Refractory Pediatric Cancers | 155 | GlobeNewswire (Europe) | Two Complete Metabolic Responses (CMRs) observed in patients with relapsed/refractory metastatic Ewing sarcoma and one Complete Response (CR) observed in a patient with relapsed/refractory metastatic... ► Artikel lesen | |
| 18.12.25 | Actuate Therapeutics: Wichtige Studiendaten zu Bauchspeicheldrüsenkrebs werden auf ASCO-GI-Symposium vorgestellt | 2 | Investing.com Deutsch | ||
| 18.12.25 | Actuate to present phase 2 pancreatic cancer study data at ASCO GI | 1 | Investing.com | ||
| 18.12.25 | Actuate Therapeutics Phase 2 Metastatic Pancreatic Cancer Data Selected for Oral and Poster Presentation at ASCO GI 2026 | 2 | GlobeNewswire (USA) | ||
| 15.12.25 | Actuate reports promising results for elraglusib in salivary cancer | 5 | Investing.com | ||
| 15.12.25 | Actuate Therapeutics Announces Publication of Positive Phase II Clinical Data for Elraglusib Combined with Platinum Chemotherapy and Sequential Immunotherapy in Recurrent, Metastatic Salivary Gland Carcinoma | 318 | GlobeNewswire (Europe) | Promising data show a median overall survival of 18.6 months, with 40% of patients alive at 2 years. Results suggest that nuclear GSK-3ß expression may help identify patients most likely to respond... ► Artikel lesen | |
| 28.11.25 | Actuate Therapeutics schließt At-the-Market-Vertriebsvereinbarung über 100 Mio. US-Dollar ab | 4 | Investing.com Deutsch | ||
| 28.11.25 | ACTUATE THERAPEUTICS, INC. - 8-K, Current Report | - | SEC Filings | ||
| 21.11.25 | ACTUATE THERAPEUTICS, INC. - 8-K, Current Report | - | SEC Filings |
1 2 3 Weiter >>
42 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 90,40 | +0,17 % | BioNTech- und Nvidia-Aktien verkauft, Medline und Alphabet rein: Erste 13F-Filings sind da! | Der britische Vermögensverwalter Baillie Gifford hat als einer der ersten sein 13F-Filing für das 4. Quartal abgegeben. Unter den großen Namen im Portfolio gibt es ein paar spannende Veränderungen.... ► Artikel lesen | |
| EVOTEC | 6,148 | +0,79 % | SDAX: Evotec-Aktie schießt zweistellig nach oben | Eine Kaufempfehlung von Berenberg entfacht neuen Optimismus bei Evotec und katapultiert die Biotech-Aktie an die Indexspitze. Die Aktie der Evotec hat am Dienstagmittag ein Kursfeuerwerk gezündet und... ► Artikel lesen | |
| BB BIOTECH | 50,60 | -0,78 % | BB Biotech: Gewinnsprung 2025 und höhere Dividende geplant | Die BB Biotech AG hat für das Geschäftsjahr 2025 vorläufig einen Nettogewinn von 578 Millionen CHF ausgewiesen, nach 76 Millionen CHF im Vorjahr. Die Ergebnisse der Biotech-Beteiligungsgesellschaft... ► Artikel lesen | |
| MEDIGENE | 0,042 | -8,26 % | Medigene: Zurückziehung - 18.11.2025 | ||
| MODERNA | 34,650 | -0,07 % | Arbutus jumps on ruling in patent dispute with Moderna | ||
| VALNEVA | 3,986 | +0,61 % | Valneva and Instituto Butantan Announce Initiation of a Pilot Vaccination Campaign in Brazil with Single-Shot Chikungunya Vaccine IXCHIQ | Lyon (France), Sao Paulo, (Brazil), February 3, 2026 -Valneva SE (Nasdaq: VALN; Euronext Paris: VLA), a specialty vaccine company and Instituto Butantan, one of the world's largest biomedical research... ► Artikel lesen | |
| NOVAVAX | 6,910 | -0,80 % | Novavax Licenses Matrix-M Adjuvant To Pfizer For Upfront Payment Of $30 Mln | NEW YORK CITY (dpa-AFX) - Novavax, Inc. (NVAX) announced Tuesday that it has entered into a license agreement with Pfizer, Inc. (PFE) for use of Novavax's Matrix-M adjuvant. Under the terms... ► Artikel lesen | |
| CRISPR THERAPEUTICS | 41,200 | -0,48 % | Morningstar, Inc.: Morningstar Completes Acquisition of CRSP and Extends Relationship with Vanguard | The CRSP integration unites two trusted sources of market insight, reinforcing a shared commitment to transparency, quality, and investor-focused solutions and solidifying Morningstar's position... ► Artikel lesen | |
| 4SC | 0,100 | -23,37 % | XFRA DELETION OF INSTRUMENTS FROM BOERSE FRANKFURT - 16.12.2025 | The following instruments on Boerse Frankfurt do have their last trading day on 16.12.2025Die folgenden Instrumente in Boerse Frankfurt haben ihren letzten Handelstag am 16.12.2025ISIN NameDE000A3E5C40 4SC... ► Artikel lesen | |
| VIVOSIM LABS | 1,220 | -100,00 % | Morning Market Movers: TechCreate, VivoSim Labs, La Rosa Holdings, Kaixin Holdings See Big Swings | BEIJING (dpa-AFX) - At 7:40 a.m. ET on Friday, premarket trading is seeing notable activity in several stocks, with early price movements signaling potential opportunities before the opening... ► Artikel lesen | |
| BIONXT SOLUTIONS | 0,387 | +2,38 % | BioNxt sichert sich innovative Chaperon-Technologie zur Verbesserung der oralen Dünnfilm-Wirkstoffverabreichung | VANCOUVER, BC / ACCESS Newswire /
5. Februar 2026 - BioNxt Solutions Inc. ("BioNxt" oder das "Unternehmen") (CSE: BNXT) (OTCQB: BNXTF) (FSE: BXT), ein biowissenschaftliches
Innovationsunternehmen... ► Artikel lesen | |
| VIKING THERAPEUTICS | 23,745 | -1,29 % | Viking Therapeutics, Inc.: Viking Therapeutics Announces Publication of Results from Phase 2 VENTURE Trial of Dual GLP-1/GIP Receptor Agonist VK2735 in the Journal Obesity | Publication Highlights Impressive Weight Loss of Up to 14.7% from Baseline After 13 Weeks of Treatment; No Plateau Observed
VK2735 Currently Being Evaluated in... ► Artikel lesen | |
| INFLARX | 0,730 | -2,54 % | Weekly Buzz: ARS Pharma's Neffy Goes East, SLS Advances, A Signal Of Hope For IFRX? | LONDON (dpa-AFX) - Despite the typical year-end slowdown, the week saw a steady stream of high-impact news, highlighted by global approvals, clinical trial readouts, new product launches, and... ► Artikel lesen | |
| BAVARIAN NORDIC | 26,050 | +0,19 % | Bavarian Nordic A/S: Bavarian Nordic - Transactions in Connection with Share Buy-Back Program | ||
| INTELLIA THERAPEUTICS | 10,320 | -0,53 % | Intellia Therapeutics, Inc.: Intellia Therapeutics Reports Inducement Grants Under Nasdaq Listing Rule 5635(c)(4) |
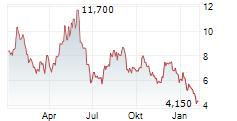
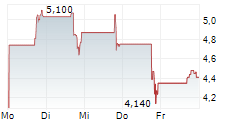
Das Unternehmen ACTUATE THERAPEUTICS INC kann der Branche Biotechnologie zugeordnet werden. Die relative Kursveränderung beträgt aktuell +1,38 % bei einem Aktienkurs von 4,410 Euro.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu ACTUATE THERAPEUTICS INC finden Sie auf Wallstreet Online.