| Realtime | Geld | Brief | Zeit |
|---|---|---|---|
| 0,033 | 0,073 | 13:04 |
- Alle
- Pressemitteilungen
- Empfehlungen
- Chartanalysen
- Berichte
| Zeit | Aktuelle Nachrichten Sprache:
Alle DE EN | Leser | Medien | ||
|---|---|---|---|---|---|
| 14.01. | Medivir AB: Bulletin From Extraordinary General Meeting In Medivir AB (publ) | 114 | GlobeNewswire (Europe) | Today on 14 January 2026, Medivir AB (publ) (the "Company") held an extraordinary general meeting in Huddinge, whereby the resolutions below were adopted.
The general meeting was held at the Company's... ► Artikel lesen | |
| MEDIVIR Aktie jetzt für 0€ handeln | |||||
| 08.12.25 | Medivir AB: Medivir's CFO Magnus Christensen to leave the company | 113 | GlobeNewswire (Europe) | Stockholm - Medivir AB (Nasdaq Stockholm: MVIR), a pharmaceutical company focused on developing innovative treatments for cancer in areas of high unmet medical need, announced today that its CFO, Magnus... ► Artikel lesen | |
| 03.12.25 | Medivir AB: Medivir announces the outcome of the Rights Issue | 174 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, HONG KONG, JAPAN, CANADA, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 27.11.25 | Medivir AB: FDA grants Medivir's MIV-711 Orphan Drug Designation for the treatment of Osteogenesis Imperfecta | 472 | GlobeNewswire (Europe) | Stockholm, Sweden - Medivir AB (Nasdaq Stockholm: MVIR), a pharmaceutical company focused on developing innovative treatments for cancer in areas of high unmet medical need, announced today that its... ► Artikel lesen | |
| 14.11.25 | Medivir AB: Medivir publishes disclosure document regarding rights issue | 176 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, HONG KONG, JAPAN, CANADA, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 11.11.25 | XFRA CAPITAL ADJUSTMENT INFORMATION - 11.11.2025 | 394 | Xetra Newsboard | Das Instrument TE5 SE0009806045 TERRANET AB B EQUITY wird ex Kapitalmassnahme gehandelt am 11.11.2025 The instrument TE5 SE0009806045 TERRANET AB B EQUITY is traded ex capital adjustment on 11.11.2025Das... ► Artikel lesen | |
| 10.11.25 | Medivir AB: Bulletin From Extraordinary General Meeting In Medivir AB (publ) | 172 | GlobeNewswire (Europe) | Today on 10 November 2025, Medivir AB (publ) (the "Company") held an extraordinary general meeting in Stockholm, whereby the resolutions below were adopted.
The general meeting was held at 7A Odenplan... ► Artikel lesen | |
| 06.11.25 | Medivir AB: Interim Report January - September 2025 | 169 | GlobeNewswire (Europe) | "With the planned study, we minimize the risks and increase the potential for fostrox to become the first approved treatment option in second-line liver cancer"
July - SeptemberFinancial summary for... ► Artikel lesen | |
| 23.10.25 | Medivir Licenses Global Rights To Topical Remetinostat To Biossil; Stock Up | 1 | RTTNews | ||
| 23.10.25 | Medivir AB: Medivir enters exclusive licensing agreement with Biossil, Inc. for remetinostat | 212 | GlobeNewswire (Europe) | Stockholm, Sweden - Medivir AB (Nasdaq Stockholm: MVIR), a pharmaceutical company focused on developing innovative treatments for cancer in areas of high unmet medical need, announces today that it... ► Artikel lesen | |
| 08.10.25 | Medivir AB: Medivir announces a fully guaranteed rights issue of approximately SEK 151 million | 186 | GlobeNewswire (Europe) | THIS PRESS RELEASE MAY NOT BE MADE PUBLIC, PUBLISHED OR DISTRIBUTED, DIRECTLY OR INDIRECTLY, IN OR INTO THE UNITED STATES OF AMERICA, AUSTRALIA, BELARUS, HONG KONG, JAPAN, CANADA, NEW ZEALAND, RUSSIA... ► Artikel lesen | |
| 21.08.25 | Medivir AB: Interim Report January - June 2025 | 177 | GlobeNewswire (Europe) | "Fostrox + Lenvima has great potential to take the place as the first approved drug treatment in second-line liver cancer"
April - JuneFinancial summary for the quarterNet turnover amounted to SEK 1.5... ► Artikel lesen | |
| 29.04.25 | Medivir AB: Interim Report January - March 2025 | 289 | GlobeNewswire (Europe) | "Final data from phase 2a study underscores potential for fostrox + Lenvima to become first approved option in second-line liver cancer"
January - MarchFinancial summary for the quarterNet turnover... ► Artikel lesen | |
| 19.03.25 | Medivir AB: Medivir receives European patent for fostrox plus lenvatinib in treatment of hepatocellular carcinoma (HCC) and cancer metastases in the liver | 270 | GlobeNewswire (Europe) | Stockholm, Sweden - Medivir AB (Nasdaq Stockholm: MVIR), a pharmaceutical company focused on developing innovative treatments for cancer in areas of high unmet medical need, announces today that the... ► Artikel lesen | |
| 25.02.25 | Medivir AB: Medivir's partner Infex Therapeutics signs a license agreement for the clinical development of MET-X in India | 246 | GlobeNewswire (Europe) | Stockholm, Sweden - Medivir AB (Nasdaq Stockholm: MVIR), a pharmaceutical company focused on developing innovative treatments for cancer in areas of high unmet medical need, announces today that its... ► Artikel lesen | |
| 18.02.25 | Medivir AB: Year-End Report January - December 2024 | 376 | GlobeNewswire (Europe) | "We are now, within short, ready to initiate the phase 2b study - with the goal of making fostrox + Lenvima the first approved medical treatment for second-line liver cancer"
October - DecemberFinancial... ► Artikel lesen |
16 Nachrichten in den letzten 12 Monaten
| Unternehmen / Aktien | Aktienkurs | % | Top-Nachrichten | ||
|---|---|---|---|---|---|
| BIONTECH | 90,40 | +0,17 % | Startschuss! BioTech-Sektor profitiert von Rotation! Augen auf bei Evotec, Bayer, Vidac Pharma and BioNTech | Mit positiver Grundstimmung hat die Börse das Jahr 2026 eingeläutet. Dass Renditechancen nicht allein im Technologiesektor liegen, bewies zuletzt der Minen- und Rohstoffbereich, wo etliche Titel Kursverdopplungen... ► Artikel lesen | |
| EVOTEC | 6,148 | +0,79 % | SDAX: Evotec-Aktie schießt zweistellig nach oben | Eine Kaufempfehlung von Berenberg entfacht neuen Optimismus bei Evotec und katapultiert die Biotech-Aktie an die Indexspitze. Die Aktie der Evotec hat am Dienstagmittag ein Kursfeuerwerk gezündet und... ► Artikel lesen | |
| BB BIOTECH | 50,60 | -0,78 % | BB Biotech: Gewinnsprung 2025 und höhere Dividende geplant | Die BB Biotech AG hat für das Geschäftsjahr 2025 vorläufig einen Nettogewinn von 578 Millionen CHF ausgewiesen, nach 76 Millionen CHF im Vorjahr. Die Ergebnisse der Biotech-Beteiligungsgesellschaft... ► Artikel lesen | |
| MEDIGENE | 0,042 | -8,26 % | Medigene: Zurückziehung - 18.11.2025 | ||
| QIAGEN | 43,170 | -0,70 % | Ionos, Kontron, Lanxess, Qiagen, Redcare Pharmacy, Siltronic, TeamViewer: Neue Aktien-Positionen der Short-Seller | Wer Aktien leerverkauft, also auf fallende Kurse spekuliert (Shortselling), unterliegt bestimmten Transparenzpflichten. Aktuelle Meldungen über Leerverkäufe geben Aufschluss darüber. Diese Vorgaben... ► Artikel lesen | |
| PAION | 0,200 | +8,11 % | XFRA MISTRADE ANTRAG IN ISIN DE000A3E5EG5 WIRD GEPRUEFT | Fuer folgende Geschaefte in der ISIN DE000A3E5EG5, Wertpapier-Name: PAION AG INH O.N., wird ein Mistrade-Antrag geprueft:ISIN Datum Zeit Volumen Preis Fair Value (AS)DE000A3E5EG5 02.02.2026 08:13:40 300 0... ► Artikel lesen | |
| VALNEVA | 3,986 | +0,61 % | Valneva and Instituto Butantan Announce Initiation of a Pilot Vaccination Campaign in Brazil with Single-Shot Chikungunya Vaccine IXCHIQ | Lyon (France), Sao Paulo, (Brazil), February 3, 2026 -Valneva SE (Nasdaq: VALN; Euronext Paris: VLA), a specialty vaccine company and Instituto Butantan, one of the world's largest biomedical research... ► Artikel lesen | |
| AMGEN | 325,15 | +0,06 % | Sanofi und Amgen: Droht diesem Ansatz das Aus? AKTIONÄR-Tipp profitiert! | Viele große Pharma- und Biotech-Firmen sind auf der Suche nach neuen Behandlungsansätzen für die Atopische Dermatitis (Neurodermitis). So auch die großen Branchenvertreter Amgen und Sanofi, die mit... ► Artikel lesen | |
| EPIGENOMICS | 0,870 | 0,00 % | PTA-Adhoc: Epigenomics AG: Vorläufiges Jahresergebnis zum 31. Dezember 2025 | DJ PTA-Adhoc: Epigenomics AG: Vorläufiges Jahresergebnis zum 31. Dezember 2025
Veröffentlichung von Insiderinformationen gemäß Artikel 17 MAR
Epigenomics AG: Vorläufiges Jahresergebnis zum... ► Artikel lesen | |
| NOVAVAX | 6,910 | -0,80 % | Novavax Licenses Matrix-M Adjuvant To Pfizer For Upfront Payment Of $30 Mln | NEW YORK CITY (dpa-AFX) - Novavax, Inc. (NVAX) announced Tuesday that it has entered into a license agreement with Pfizer, Inc. (PFE) for use of Novavax's Matrix-M adjuvant. Under the terms... ► Artikel lesen | |
| BIOGEN | 164,25 | +3,79 % | Ihre wichtigsten Termine: Frische Zahlen von Philip Morris, Under Armour, Toyota, Biogen, Societe Generale | © Foto: Hiro Komae/AP/dpaGut geplant in den Tag. Mit dem wO-Tagesausblick haben Sie die wichtigsten Termine im Blick und starten bestens vorbereitet in den neuen Handelstag.Unternehmenstermine 06:30... ► Artikel lesen | |
| BIOFRONTERA | 2,420 | -1,22 % | PTA-news: Biofrontera AG: Biofrontera berichtet über die Ergebnisse der ersten neun Monate des Jahres 2025 | DJ PTA-News: Biofrontera AG: BIOFRONTERA BERICHTET ÜBER DIE ERGEBNISSE DER ERSTEN NEUN MONATE DES JAHRES 2025
Unternehmensmitteilung für den Kapitalmarkt
Biofrontera AG: BIOFRONTERA... ► Artikel lesen | |
| HEIDELBERG PHARMA | 2,910 | 0,00 % | HEIDELBERG PHARMA AG zeigt klare Stärke im Trend | ||
| ILLUMINA | 101,04 | -0,18 % | Illumina-Aktie -10%: Kann man hier günstig einsteigen? | Mit einem Kursverlust von -10% war die Illumina-Aktie am Freitag der große Verlierer im S&P 500. Der Hersteller von Gensequenzierungsmaschinen setzte damit seinen Abwärtstrend der letzten Tage fort.... ► Artikel lesen | |
| NANOREPRO | 1,535 | +2,68 % | NanoRepro ordnet Aufsichtsrat nach außerordentlicher HV neu | Die Aktionäre der NanoRepro AG haben auf der außerordentlichen Hauptversammlung vom 16. Januar 2026 den Aufsichtsrat neu zusammengesetzt. Neu gewählt wurden Gunter Heneis, Michael Herrmann und Konstantin... ► Artikel lesen |
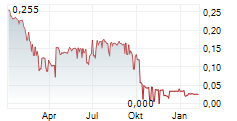
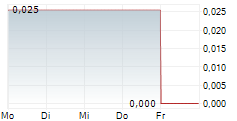
Das Unternehmen MEDIVIR AB kann der Branche Biotechnologie zugeordnet werden. Der aktuelle Kurs liegt bei 0,033 Euro und damit +28,35 % im Plus.
VerkaufenHaltenKaufen
■ Verkaufen
■ Halten
■ Kaufen
Sie erhalten auf FinanzNachrichten.de kostenlose Realtime-Aktienkurse von und sowie Kurse und Daten von ARIVA.DE AG.
Weitere Kennzahlen, Fundamentaldaten und Unternehmensinformationen zu MEDIVIR AB finden Sie auf Wallstreet Online.